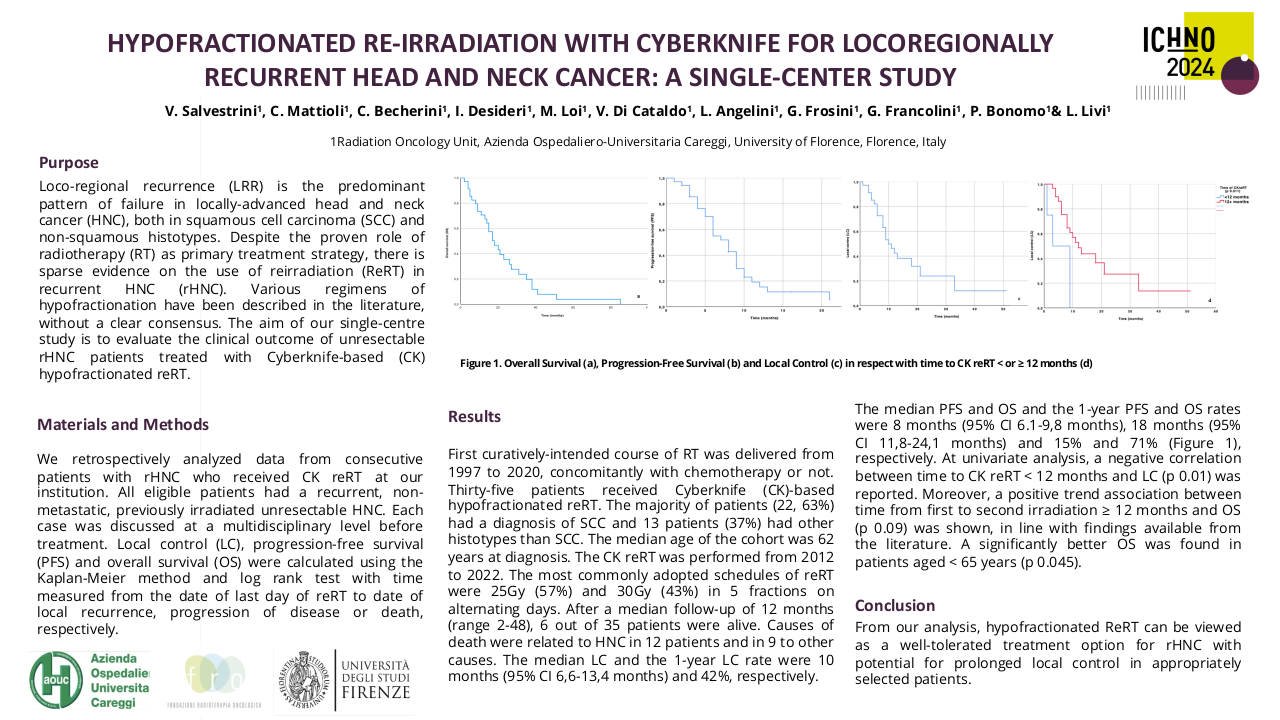
Evolution of phase 1 clinical trials for head and neck cancer patients based on the experience of a dedicated phase I Unit.
Purpose/Objective
Since the approval of anti-EGFR and anti-PD1 monoclonal antibodies in head and neck squamous cell carcinoma, there is an increased interest in developing novel therapeutics in head and neck malignancies. The purpose of this study is to analyze all phase I clinical trials conducted at the Vall d’Hebron Institute of Oncology for head and neck cancers since 2010. We aimed to evaluate the proportion of phase I studies with dedicated cohorts for head and neck cancers, assessing the types of agents in development and tumor types included, as well as analyzing the patient characteristics participating in these trials.
Material/Methods
We have reviewed all the phase 1 clinical trials conducted in the Early Drug Development Unit, from Vall d´Hebron Institute of Oncology from January 2010 to June 2023, and identify which of these studies have cohorts for head and neck cancers. We use descriptive statistics to describe the tumor types and drug types included in these cohorts. We have also collected data from all the patients with head and neck cancers, included in phase I clinical trials from January 2010 to December 2022 and describe tumor types and agents used to identify potential opportunities for development in head and neck tumors.
Results
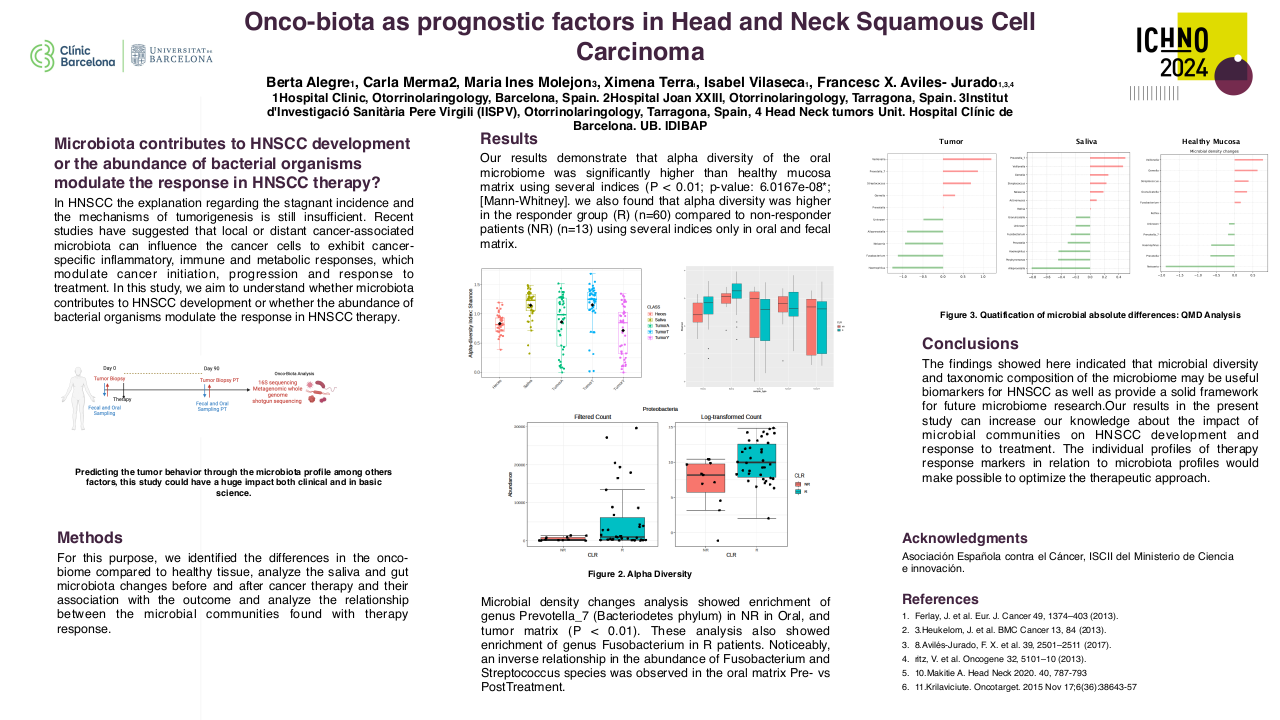
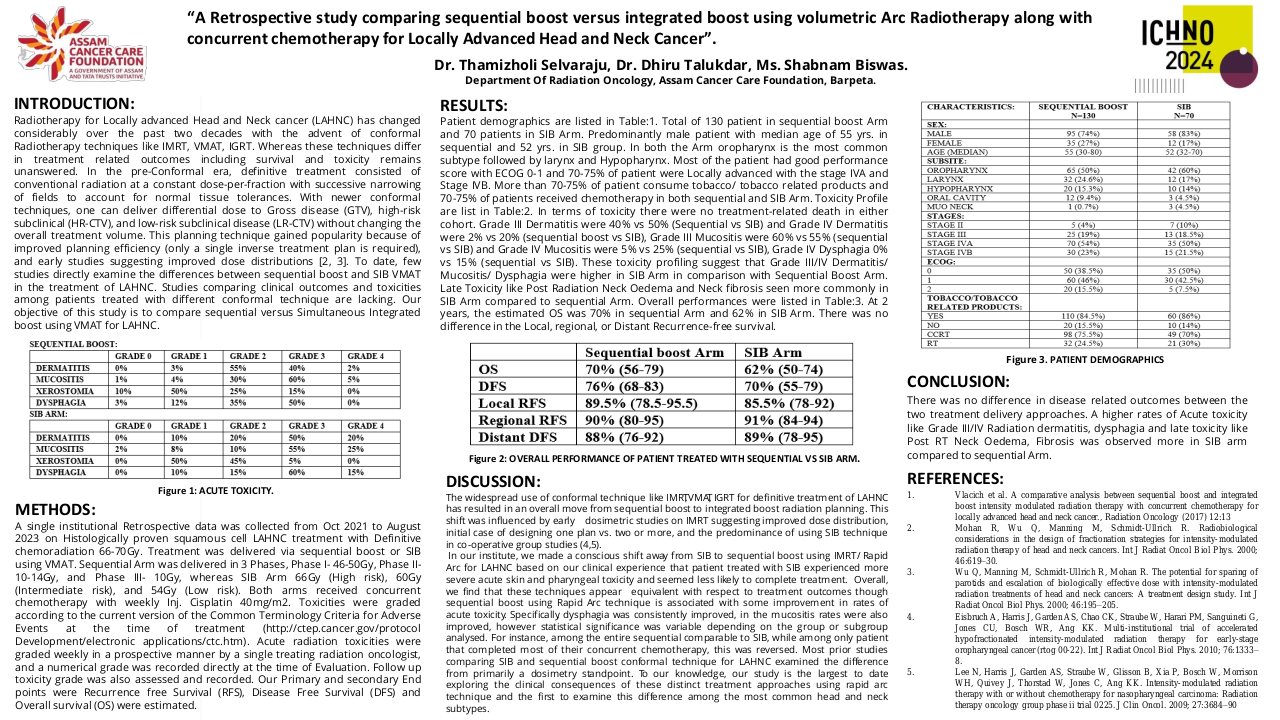
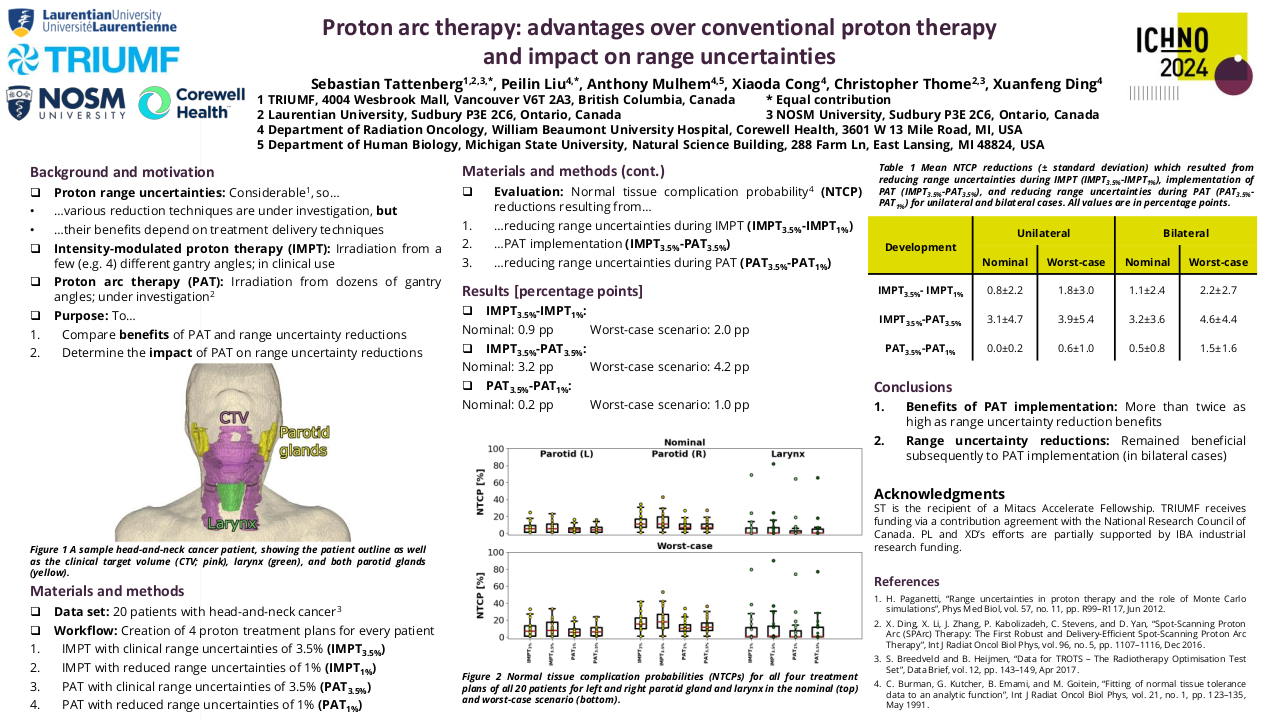
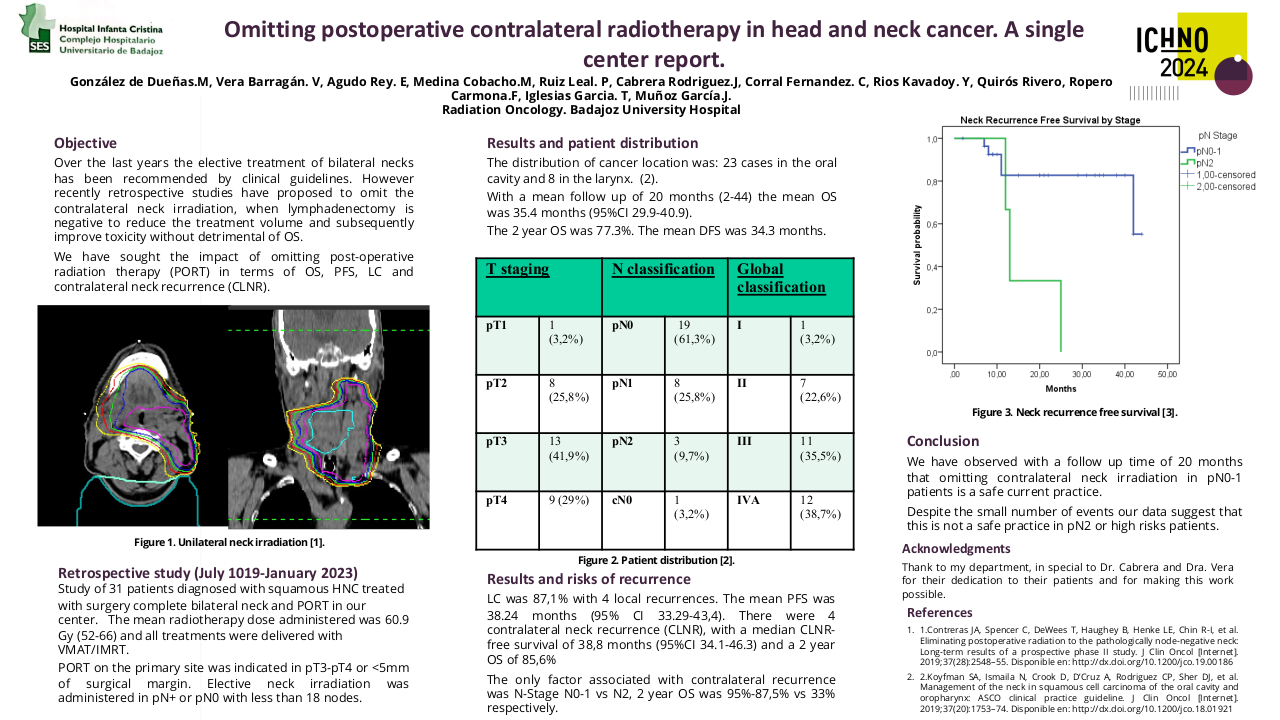
Among the 634 phase I clinical trials activated from 2010 to 2023, 151 clinical trials had at least one cohort for patients with head and neck malignancies. Most of these studies include other solid tumors, and only 2 studies were tumor-specific. The majority of the 151 studies with dedicated head and neck cohorts were focused on head and neck squamous cell carcinoma (84%), while only 21% and 2.8% included salivary gland cancers or nasopharyngeal carcinomas (7.5% of studies include two or more histologies). Sixty-two studies evaluated agents in monotherapy, while 38% evaluated combinations. The anticancer agents evaluated included: targeted therapy 131 (87%), immunotherapy 61 (40%), chemotherapy 17 (11%), and hormone therapy 1 (0.7%). The proportion of studies evaluating immunotherapies was higher in the studies with dedicated head and neck cohorts, in comparison to the global pool of phase I studies conducted in the unit (32.5%). Although the absolute number of phase I studies with dedicated head and neck cohorts has progressively increased (29 studies in 2022), the proportion compared to the global pool of phase I studies remained stable since 2016 ranging from 25-30% of the total.
From January 2010 to December 2022 we have included 275 patients in phase I clinical trials: 63% had HNSCC, 28% had salivary gland cancers, 8% had nasopharyngeal carcinomas, and 1% had other tumor types. The new agents evaluated included: immunotherapies (52%), targeted agents (41%), novel chemotherapies or antibody-drug conjugates (6%), and other therapies such as nanoparticles (1%).
Conclusion
There is an increasing interest in developing novel drugs for head and neck cancers. In recent years, 25-30% of the phase clinical trials conducted in our institution have at least one cohort dedicated to head and neck malignancies, especially HNSCC. Despite these positive trends, clinical trial opportunities for rare/less frequent head and neck cancers remain limited.